 (2012, December 18) Valence Electrons and the Periodic Table. If the valence shell of an element is full, such as with a noble gas, then the element does not want to gain or lose an electron.įor example, alkali metals, which all have a valency of 1, want to lose that one electron and are likely to form ionic bonds (such as in the case of NaCl, or table salt) with a Group 17 element, which has a valency of 7 and wants to gain that one electron from the alkali metal (Group 1 element) to form a stable valency of 8.įor more on valence electrons and how they're related to the periodic table, I strongly recommend this video:Ĭitations: Tyler Dewitt. The electron configurations of these 30 elements are unique due to their valence electrons being located in the 4f and 5f sublevels, which does not occur on any other elements on the table. They determine how "willing" the elements are to bond with each other to form new compounds. Valence is typically the number of electrons needed to fill the outermost shell of an atom. Valence electrons are responsible for the reactivity of an element. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table.įor example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively.Ītoms in Groups 13 and 18 have 3 and 8 valence electrons, respectively. Valence electrons are the electrons present in the outermost shell of an atom. To form a covalent bond, one electron from the halogen and one electron from another atom form a shared pair.įor example, in #"H–F"#, the dash represents a shared pair of valence electrons, one from #"H"# and one from #"F"#. Elements which have eight valence electrons ( noble gases) are inert and they do not tend to create chemical. group: A vertical column in the periodic table which signifies the number of valence shell electrons in an elements atom. As a gas or vapor, the halogens all had a pungent odor. They determine the valency of the atom which is important in how a chemical element reacts with other elements. To form an ionic bond, a halogen atom can remove an electron from another atom in order to form an anion (e.g., #"F"^"-", "Cl"^"-"#, etc.). In chemistry, valence electrons are the electrons in the outside or valence electron shell of an atom. 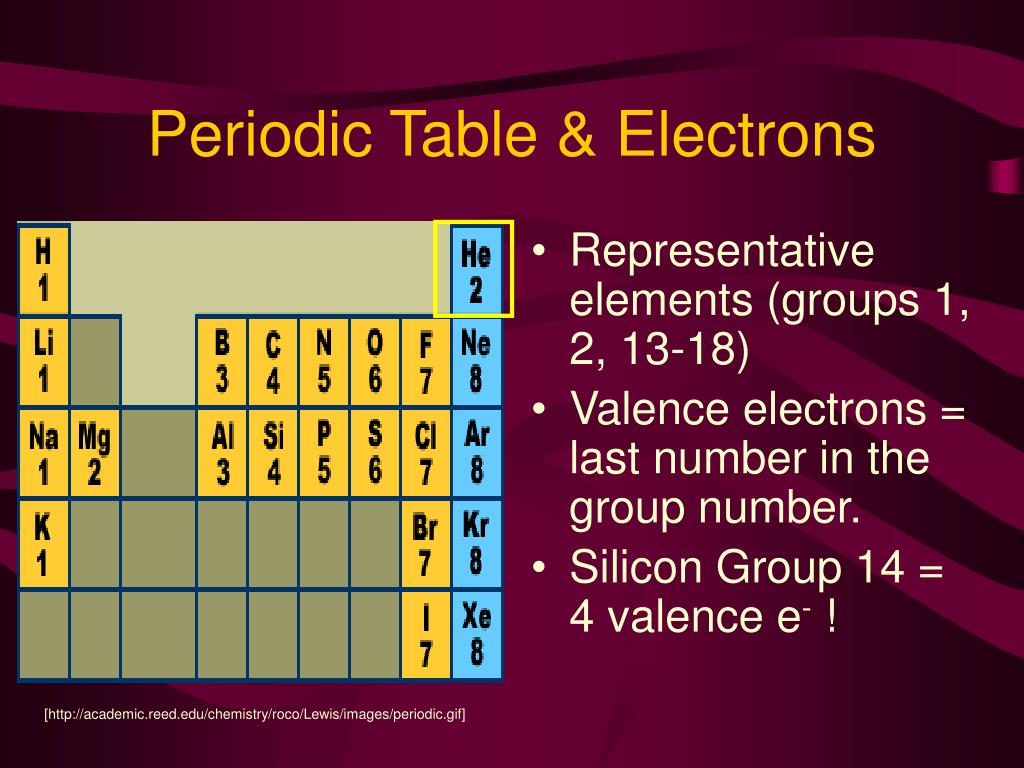 
They have one less electron configuration than a noble gas, so they require only one additional valence electron gain an octet. The most reactive nonmetals are the halogens, e.g., #"F"# and #"Cl"#. Only the noble gases (the elements on the right-most column of the periodic table) have zero charge with filled valence. In a single covalent bond, a shared pair forms with both atoms in the bond each contributing one valence electron. For most atoms there will be a maximum of eight electrons in the valence shell (octet structure), e.g., \(CH4\) Figure 1: Bonding in \(H2\) and methane (\(CH4\)) The other tendency of atoms is to maintain a neutral charge. Nonmetals tend to attract additional valence electrons to form either ionic or covalent bonds. In chemistry and physics, valence electrons are electrons in the outermost shell of an atom, and that can participate in the formation of a chemical bond if the outermost shell is not closed. They need to lose only one or two valence electrons to form positive ions with a noble gas configuration. The most reactive metals are those from Groups 1 and 2. Generally, elements in Groups 1, 2, and 13 to 17 tend to react to form a closed shell with a noble gas electron configuration ending in #ns^2 np^6#. However, these elements are reactive enough that they do not exist in their elemental forms in nature, but are present as compounds.Elements whose atoms have the same number of valence electrons are grouped together in the Periodic Table. The need to remove two electrons in order for the material to react means more energy is needed for electron removal. Look on the periodic table to see what group the element is in. 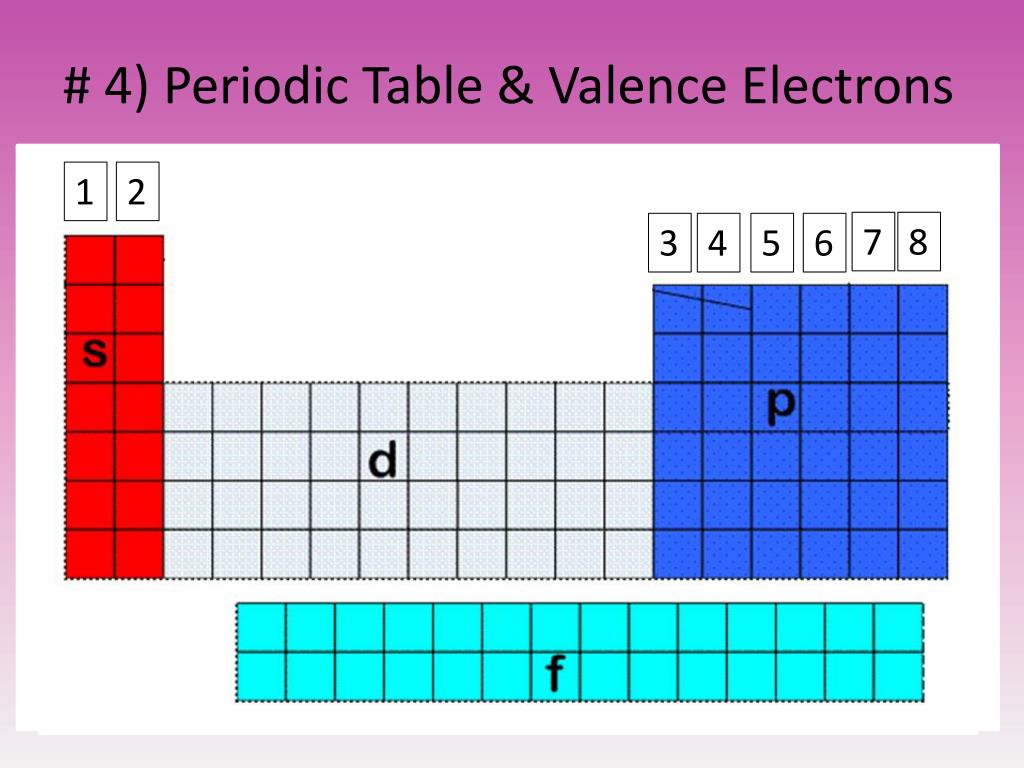
The second shell has 2 subshells: 1 s-orbital and 3 p-orbitals. 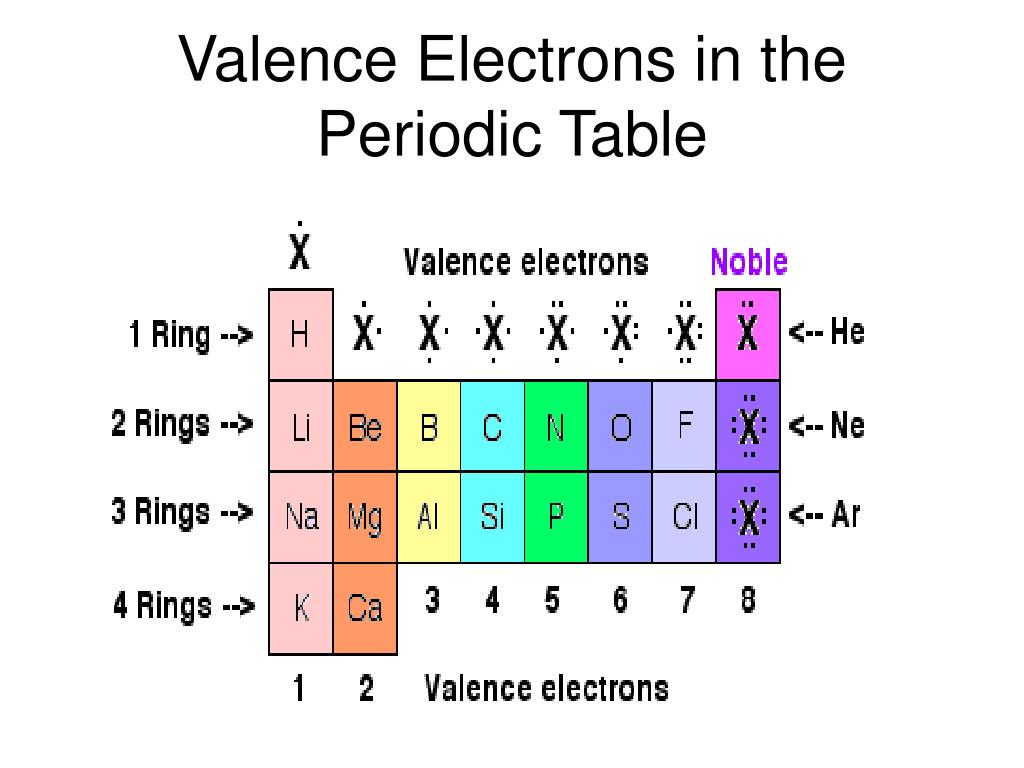
This means that the first shell can hold 2 electrons. The first shell (of all atoms) has 1 subshell of s-orbitals containing 1 s orbital. The Group 2 elements tend to be less reactive than their Group 1 counterparts. We only use the valence electrons because those are the only ones involved in chemical bonding. Every subshell has a of orbits s/p/d/f that can each hold 2 electrons each (one has the opposite spin of the other). Whether you're supplementing in-class learning or assigning. However, radium is a radioactive element and is generally under the category of radioisotopes in addition to being an alkaline earth metal, because it is not a stable element. Browse our collection of Texas Standards-aligned chemistry practice problems, step-by-step skill explanations, and video walkthroughs. Radium (atomic number 88) has similar properties to barium and is also in the Group 2 category. \) (Credit: Ingmar Runge Source: (opens in new window) License: Public Domain) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
